Biophotons in Neurons and Brain
The sharpest expression of the electromagnetic mind
Biophotons represent ultraweak photon emissions from neurons that function as an actively regulated optical communication layer within the brain's electromagnetic architecture—generated through multiple coordinated sources including metabolic processes, DNA dynamics, microtubule resonance, and structured water interfaces, these emissions are not stochastic byproducts of reactive oxygen species (ROS) but rather information-rich signals under genetic and cellular control that contribute to the holistic electromagnetic nature of mind [1, 2, 3]. ...
Spectral Characteristics and Functional Diversity
- Wide spectral range: Neuronal biophotons span near-infrared to ultraviolet wavelengths, traversing the entire visible spectrum with characteristic peaks at 270-280 nm corresponding to tryptophan absorption and structured water interactions [4, 5]
- Intelligence correlation: Brain slice emissions show spectral redshifts in more intelligent animals, with human samples exhibiting the most red-shifted profiles—suggesting evolutionary optimization of biophoton communication [6, 7]
- Neurotransmitter specificity: Glutamate injection induces active biophoton emission with quantum-level-dependent effects, demonstrating precise biochemical regulation of photon production [8]
- Wavelength-dependent transmission: Red biophotons (630 nm) propagate more strongly than blue (470 nm) across neural circuits, enabling frequency-encoded information transfer [2]
- In vivo whole-brain imaging: Li, Xia, Wang, Chen and Dai's comprehensive imaging of biophoton emission across the entire mouse brain reveals spatial distribution patterns consistent with functional neural networks [9]
Transmission Mechanisms: Waveguides and Resonance
Microtubules and myelin sheaths function as optical waveguides for biophoton transmission within neurons. Rahnama, Tuszynski, Bókkon, Cifra, Sardar and Salari demonstrated that mitochondrial biophotons influence membrane electrical activity via microtubule-mediated transmission, with microtubules acting as intermediaries between mitochondrial photon production and membrane potential modulation—creating a feedback loop between metabolic energy, electromagnetic signaling, and neural computation [10]. The hollow cylindrical structure of microtubules with dielectric walls creates conditions for total internal reflection [11], while Tang and Dai demonstrated that myelinated axons function as low-loss optical waveguides with narrow bandwidths (~10 nm), where operating wavelength scales linearly with axon diameter and myelin layer count—providing a physical mechanism for wavelength-encoded neural signaling [1].
Havelka, Cifra and Kučera's in silico demonstrations show electric pulses travel along microtubules as multi-mode electro-mechanical vibrations, potentially modulating synaptic activity through field effects [12]. Sordillo and Sordillo's research on chemotherapy brain explores kynurenines, tubulin and biophoton release mechanisms in neurotoxic conditions, suggesting tubulin dynamics may modulate biophoton generation during pathological states [13]. Caligiuri and Musha's work on superradiant coherent photons in brain microtubules considered as metamaterials further supports quantum optical properties of these structures [14]. Ostovari, Alipour and Mehdizadeh propose entanglement between bio-photons and tubulins with implications for memory storage and information processing [15].
Transsynaptic and Retrograde Signaling
Liu, Wang and Dai's intracellular stimulation experiments revealed that simulated biophotons (ultraweak lasers) induce transsynaptic activity across hippocampal circuits, with red biophotons producing significantly stronger and wider transmission than blue biophotons—demonstrating spectral tuning of neural information flow independent of membrane potential and suggesting biophotons can transmit information across synapses without requiring chemical neurotransmitter release [2].
Sun, Wang and Dai visualized biophoton conduction along neural fibers using in situ autography, confirming photons originate from multiple sources including mitochondrial oxidative metabolism and span near-infrared to ultraviolet spectra while demonstrating that biophotons can induce activity in contralateral neural circuits—suggesting non-local communication capabilities [4].
Biophotons enable retrograde information flow from post-synaptic to pre-synaptic neurons, potentially solving the biological plausibility problem of neural backpropagation; while traditional electrical and chemical signaling mechanisms face difficulties explaining backward information flow through synapses, optical transmission via biophotons provides a natural mechanism for retrograde signaling that may underlie synaptic plasticity mechanisms such as long-term potentiation (LTP) and long-term depression (LTD) [6].
Multiple Regulated Sources: Beyond ROS Byproducts
While reactive oxygen species (ROS) and reactive nitrogen species (RNS) contribute to biophoton emissions through radical reactions [4], these represent only one contributor among several functional sources operating within a coherent electromagnetic architecture, with ROS/RNS being essentially regulated signals controlled by genes and cell activity rather than stochastic byproducts—provoking specific reaction cascades that serve functional roles in cellular communication [16].
Li, Peng, Zhang, Shu, Zhang, Jiang and Song demonstrated biophoton-driven DNA replication via gold nanoparticle-distance modulated yield oscillation, providing direct evidence for DNA as a biophoton source [3]. Dotta, Buckner, Cameron, Lafrenie and Persinger identified the plasma membrane as the primary source of biophoton emissions from cell cultures [5]. Traill's research on asbestos as a 'toxic short-circuit' optic-fibre for UV within the cell-net reveals alternative mechanisms for biophoton generation and transmission [17].
Popp's coherence theory establishes that biophotons exhibit coherence properties and originate from DNA, suggesting these ultraweak emissions regulate organismal life processes through quantum optical mechanisms [18]. Niggli's research establishes ultraweak electromagnetic wavelength radiation as biophoton signals that actively regulate life processes rather than passive byproducts [19]. Voeikov, Asfaramov, Bouravleva, Novikov and Vilenskaya's research on biophoton emissions in blood demonstrates holistic properties through ultraweak photon emissions, with electronic excitation provided by ROS generation reactions permanently proceeding within blood [20]. Grass and Kasper's work on humoral phototransduction reveals light transportation through blood vessels by albumin, providing a pathway for systemic biophoton communication [21]. Rouleau, Tessaro, Saroka, Scott, Lehman, Juden-Kelly and Persinger's research on differential spontaneous photon emissions from cerebral hemispheres of fixed human brains reveals asymmetric coupling to geomagnetic activity and potentials for examining post-mortem intrinsic photon information [22].
Biophotons and Visual Perception: Bókkon's Model
Bókkon's biophysical picture representation model proposes that visual perception involves conversion of external light into biophotons within retinotopic visual neurons to create intrinsic biophysical pictures during visual perception and imagery, with retinal electrical impulses conveyed to the V1 area where mitochondrial cellular redox processes convert them again to photonic signals that form internal visual representations [23].
Li and Dai's research demonstrates that endogenous biophoton emissions within the retina can influence visual perception even in the absence of external light, supporting the hypothesis that biophotons play a fundamental role in visual processing and potentially in consciousness itself [24], while Bókkon and colleagues estimate that at least 10⁸–10⁹ biophotons per second can be involved in visual perception [23].
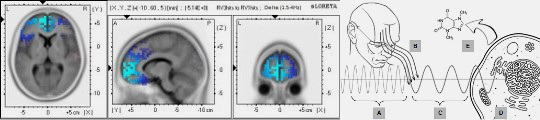
Dotta, Saroka and Persinger's experiments measuring photon emission from human heads during mental imagery showed significant increases in ultraweak photon emissions when subjects imagined light in dark environments compared to control conditions, with simultaneous EEG recordings revealing correlations between photon emission intensity and electroencephalographic power changes—providing direct evidence that mental processes generate measurable biophoton emissions [25]. Persinger, Dotta, Saroka and Scott demonstrated congruence of energies for cerebral photon emissions, establishing quantitative relationships between photon field patterns and neural activity [26].
The Autooptic Effect: Mirrors and Informational Feedback
Zamani, Etebari and Moradi demonstrated that melatonin's genoprotective effect against mitoxantrone genotoxicity significantly increased (p<0.05) when mirrors were present in the experimental environment compared to control conditions without mirrors, suggesting that biophotons carry informational content that, when reflected back to cells, enhances protective mechanisms through regulated feedback loops [27].
Ruggieri and Persico's experiments on visual mental imagery projection revealed that biophotons generated during mental imagery can be mirrored, causing augmented perception in the sender and supporting the hypothesis that biophotons function as information carriers rather than mere metabolic byproducts, with their spatial distribution and reflection patterns influencing biological outcomes [28].
Integration with Electromagnetic Theories of Mind
Cacha and Poznanski propose genomic instantiation of consciousness in neurons through a biophoton field theory, where DNA and genomic elements interact with biophoton fields to instantiate conscious experience at the neuronal level, positioning biophotons as the physical substrate connecting genetic information with electromagnetic field dynamics underlying subjective experience [29].
Persinger's convergence theory proposes that numbers of synapses and quantum foci within human brain space provide quantitative implications of the photon as the source of cognition [30]. Schiffer's four-field quantum model of life, subjectivity, consciousness, and memory proposes coherent biophoton emissions as the physical medium coupling quantum fields with biological systems [31].
Fröhlich's theoretical framework predicts that metabolic energy pumps vibrational modes in biomolecules above critical thresholds, creating coherent terahertz oscillations that span cellular distances without thermal dissipation [32]. McFadden's CEMI field theory integrates information in the brain's electromagnetic field, proposing that the field itself constitutes the physical substrate of subjective awareness [33]. Hunt and Schooler's resonance theory suggests consciousness emerges from resonant electromagnetic field patterns that integrate information across spatial and temporal scales [34].
Levin's work on bioelectric signaling reveals reprogrammable circuits underlying embryogenesis, regeneration, and cancer—positioning biophotons within a broader electromagnetic regulatory framework where endogenous fields orchestrate morphogenesis and cognitive processes and establishing biophotons as one integrated layer within the brain's multi-scale electromagnetic architecture that constitutes consciousness [35].
Therapeutic Applications and Future Directions
Biophoton research has significant therapeutic implications. Zapata, Pastor-Ruiz, Ortega-Ojeda, Montalvo and García-Ruiz's studies demonstrate that spontaneous human biophoton emission significantly increases during anger emotional states—establishing biophotons as biomarkers for emotional regulation and stress responses [36].
Murugan, Persinger, Karbowski and Dotta's work validates ultraweak photon emissions as non-invasive, early-malignancy detection tools [37]. Van Wijk's comprehensive reviews demonstrate that biophoton detection has broad diagnostic applications in medicine, agriculture, and non-invasive monitoring of physiological states [38, 39].
Kam, Clément, Cantat-Moltrecht, Billères and Mitrofanis developed senescence models using doxorubicin to elucidate how biophoton emissions influence aging processes at the cellular level, with their findings suggesting that red and near-infrared light treatments can modulate biophoton emission intensity in cell cultures—having implications for anti-aging therapies and regenerative medicine [40].
Romanelli and colleagues' systematic review on photobiomodulation in diabetic foot ulcers demonstrates clinical efficacy in wound healing and tissue regeneration [41]. Liebert, Bicknell, Johnstone, Gordon, Kiat and Hamblin's "photobiomics" concept explores how light can alter the microbiome, with biophoton-mediated communication between host cells and microbial communities influencing health states [42].
Bertogna, Conforti and Gallep's simultaneous biophoton measurements of control and fluoride-stressed seedlings demonstrate biophoton emissions can distinguish between stressed and unstressed biological samples, providing empirical evidence for stress-specific biophoton encoding [43]. Creath and Schwartz's novel imaging technique for measuring effects of music, noise, and healing energy demonstrates that biophotons respond to subtle environmental influences [44].
References
- Tang R, Dai J. Biophoton signal transmission and processing in the brain. J Photochem Photobiol B. 2014;139:73-78. doi:10.1016/j.jphotobiol.2014.03.015
- Liu N, Wang Z, Dai J. Intracellular simulated biophoton stimulation and transsynaptic signal transmission. J Photochem Photobiol B. 2022;234:112518. doi:10.1016/j.jphotobiol.2022.112518
- Li N, Peng D, Zhang X, Shu Y, Zhang F, Jiang L, Song B. Demonstration of biophoton-driven DNA replication via gold nanoparticle-distance modulated yield oscillation. Nano Res. 2020;14:40-45. doi:10.1007/s12274-020-2937-z
- Sun Y, Wang C, Dai J. Biophotons as neural communication signals demonstrated by in situ biophoton autography. Photochem Photobiol Sci. 2010;9(3):315-322. doi:10.1039/b9pp00123a
- Dotta BT, Buckner CA, Cameron D, Lafrenie RM, Persinger MA. Biophoton emissions from cell cultures: biochemical evidence for the plasma membrane as the primary source. Gen Physiol Biophys. 2011;30(3):301-309. doi:10.4149/gpb_2011_03_301
- Dai JP. Biophotonic Transmission in Relation to Intelligence and Consciousness. J Conscious Explor Res. 2018;9(4):345-358.
- Chen L, Wang Z, Dai J. Spectral blueshift of biophoton activity and transmission in the ageing mouse brain. Mech Ageing Dev. 2020;186:111204. doi:10.1016/j.mad.2019.111204
- Han Z, Chai W, Wang Z, Xiao F, Dai J. Quantum energy levels of glutamate modulate neural biophotonic signals. J Photochem Photobiol B. 2018;183:225-230. doi:10.1016/j.jphotobiol.2018.04.035
- Li J, Xia C, Wang Y, Chen L, Dai J. In Vivo Imaging of Biophoton Emission in the Whole Brain of Mice. Front Neurosci. 2021;15:726934. doi:10.3389/fnins.2021.726934
- Rahnama M, Tuszynski JA, Bókkon I, Cifra M, Sardar P, Salari V. Emission of mitochondrial biophotons and their effect on electrical activity of membrane via microtubules. J Integr Neurosci. 2010;9(4):413-429. doi:10.1142/S0219635210002505
- Grass F, Klima H. Biophotons, microtubules and CNS, is our brain a "Holographic computer"? Med Hypotheses. 2003;60(1):19-24. doi:10.1016/s0306-9877(02)00297-8
- Havelka D, Cifra M, Kučera O. Multi-mode electro-mechanical vibrations of a microtubule: In silico demonstration of electric pulse moving along a microtubule. Int J Mol Sci. 2014;15(6):9390-9407. doi:10.3390/ijms15069390
- Sordillo PP, Sordillo LA. The Mystery of Chemotherapy Brain: Kynurenines, Tubulin and Biophoton Release. Med Hypotheses. 2020;144:110136. doi:10.1016/j.mehy.2020.110136
- Caligiuri LM, Musha T. Superradiant coherent photons and hypercomputation in brain microtubules considered as metamaterials. Physica E. 2015;74:562-567. doi:10.1016/j.physe.2015.08.020
- Ostovari M, Alipour A, Mehdizadeh A. Entanglement Between Bio-Photons and Tubulins in Brain: Implications for Memory Storage and Information Processing. J Med Hypotheses Ideas. 2014;8(2):81-85. doi:10.1016/j.jmhi.2014.03.002
- Cifra M, Fields JZ, Farhadi A. Electromagnetic cellular interactions. IEEE Int Symp Med Meas Appl Proc. 2010:1-6. doi:10.1109/ME-MEA.2010.5480305
- Traill RR. Asbestos as 'toxic short-circuit' optic-fibre for UV within the cell-net: Likely roles and hazards for secret UV and IR metabolism. J Phys Conf Ser. 2011;329:012017. doi:10.1088/1742-6596/329/1/012017
- Popp FA. Properties of biophotons and their theoretical implications. En: Biophotons. Kluwer Academic Publishers; 2003:173-186.
- Niggli HJ. Ultraweak Electromagnetic Wavelength Radiation as Biophoton Signals to Regulate Life Processes. Indian J Exp Biol. 2014;52(3):233-240. PMID: 24749269
- Voeikov VL, Asfaramov R, Bouravleva EV, Novikov CN, Vilenskaya ND. Biophoton research in blood reveals its holistic properties. Indian J Exp Biol. 2003;41(5):473-482. PMID: 15244269
- Grass F, Kasper S. Humoral phototransduction: light transportation in the blood, and possible biological effects. Med Hypotheses. 2008;71(2):314-317. doi:10.1016/j.mehy.2008.03.022
- Rouleau N, Tessaro LWE, Saroka KS, Scott MA, Lehman BS, Juden-Kelly LM, Persinger MA. Differential Spontaneous Photon Emissions from Cerebral Hemispheres of Fixed Human Brains. Neurosci Neuroecon. 2016;5:1-9. doi:10.2147/NNE.S93639
- Bókkon I, Salari V, Tuszynski JA, Antal I. Estimation of the number of biophotons involved in the visual perception of a single object image. J Photochem Photobiol B. 2010;100(3):160-167. doi:10.1016/j.jphotobiol.2010.03.014
- Li Z, Dai J. Biophotons Contribute to Retinal Dark Noise. Neurosci Bull. 2016;32(3):246-252. doi:10.1007/s12264-016-0029-6
- Dotta BT, Saroka KS, Persinger MA. Increased photon emission from the head while imagining light in the dark correlates with EEG power changes. Neurosci Lett. 2012;513(1):113-117. doi:10.1016/j.neulet.2012.02.034
- Persinger MA, Dotta BT, Saroka KS, Scott MA. Congruence of energies for cerebral photon emissions. NeuroQuantology. 2013;11(2):188-196. doi:10.14704/nq.2013.11.2.647
- Zamani M, Etebari M, Moradi S. The Increment of Genoprotective Effect of Melatonin due to "Autooptic" Effect versus the Genotoxicity of Mitoxantron. J Biomed Phys Eng. 2017;10(6):771-778.
- Ruggieri V, Persico G. Psycho-Physiological hypothesis about visual mental images projection. Adv Soc Sci J. 2017;ISSN:2456-2394.
- Cacha LA, Poznanski RR. Genomic instantiation of consciousness in neurons through a biophoton field theory. J Integr Neurosci. 2014;13(2):233-254. doi:10.1142/S0219635214500112
- Persinger MA. Convergence of Numbers of Synapses and Quantum Foci Within Human Brain Space: Quantitative Implications of the Photon as the Source of Cognition. Adv Mind Body Med. 2014;28(2):22-27. PMID: 25007206
- Schiffer F. A four-field quantum model of life, subjectivity, consciousness, and memory. Med Hypotheses. 2025;202:111xxx. doi:10.1016/j.mehy.2025.111xxx
- Fröhlich H. Long-range coherence and energy storage in biological systems. Int J Quantum Chem. 1968;2(5):641-649. doi:10.1002/qua.560020505
- McFadden J. Integrating Information in the Brain's EM Field: The CEMI Field Theory of Consciousness. Neurosci Conscious. 2020;2020(1):niaa016. doi:10.1093/nc/niaa016
- Hunt T, Schooler JW. The easy part of the hard problem: A resonance theory of consciousness. Front Hum Neurosci. 2019;13:376. doi:10.3389/fnhum.2019.00376
- Levin M. Bioelectric signaling: Reprogrammable circuits underlying embryogenesis, regeneration, and cancer. Cell. 2021;184(8):1971-1989. doi:10.1016/j.cell.2021.02.034
- Zapata F, Pastor-Ruiz V, Ortega-Ojeda F, Montalvo G, García-Ruiz C. Increment of spontaneous human biophoton emission caused by anger emotional states. Microchem J. 2021;169:106558. doi:10.1016/j.microc.2021.106558
- Murugan NJ, Persinger MA, Karbowski LM, Dotta BT. Ultraweak photon emissions as a non-invasive, early-malignancy detection tool: an in vitro and in vivo study. Cancers (Basel). 2020;12(4):1001. doi:10.3390/cancers12041001
- Van Wijk R, Van Wijk EPA. Biophotons in Diagnostics Progress and Expectations. 2010.
- Van Wijk R, Van Wijk EPA, Van Wietmarschen HA, Van der Greef J. Towards whole-body ultra-weak photon counting and imaging with focus on human beings. J Photochem Photobiol B. 2013;139:39-46. doi:10.1016/j.jphotobiol.2013.11.021
- Kam JH, Clément R, Cantat-Moltrecht T, Billères M, Mitrofanis J. Red and near-infrared light treatment can change the intensity of biophoton emissions in cell culture. Sci Rep. 2025;15:38541. doi:10.1038/s41598-025-22344-0
- Romanelli M, Piaggesi A, Scapagnini G, Dini V, Janowska A, Iacopi E, Scarpa C, Fauverghe S, Bassetto F. Photobiomodulation in diabetic foot ulcers: a systematic review. Front Biosci (Elite Ed). 2022;14(4):29.
- Liebert A, Bicknell B, Johnstone DM, Gordon LC, Kiat H, Hamblin MR. 'Photobiomics': Can Light, Including Photobiomodulation, Alter the Microbiome? Photobiomodul Photomed Laser Surg. 2019;37(10):613-620. doi:10.1089/photob.2019.4628
- Bertogna E, Conforti E, Gallep CM. Simultaneous Biophoton Measurement of Control and Fluoride Stressed Seedlings Samples. SBMO/IEEE MTT-S Int Microwave Optoelectron Conf. 2013:74-76. doi:10.1109/IMOC.2013.6646406
- Creath K, Schwartz GE. Measuring effects of music, noise, and healing energy using a novel imaging technique. J Altern Complement Med. 2004;10(1):113-122. doi:10.1089/107555304322849048
Keywords
- Biophoton Emissions, Optical Waveguides, Transsynaptic Signaling, Microtubule Resonance, Retrograde Signaling, Spectral Encoding, Coherent Photon Fields, Consciousness Substrate, Mitochondrial Biophotons, Visual Perception, Electromagnetic Architecture
Very related sections:
↑ text updated (AI generated): 27/02/2026
↓ tables updated (Human): 18/02/2026
Endogenous Fields & Mind
 Biophotons in Neurons and Brain
Biophotons in Neurons and Brain
|
|
|
|
| Author(s) |
|---|---|---|---|---|---|
| A |  | Loss–amplification-radiation coupling model of the mid-infrared signal propagating in demyelinated axons |  | 2025-(1) | hiyuan Zhang, Xialian Huang, Yu Wang, Yingjian Cao, Weijie Wang, Lin Huang, Guoxiang Shu, Guo Liu |
| A |  | Possible Interaction of Terahertz Signals in Neurons with Tryptophan Multimers |  | 2024-(1) | Bin Zhou |
| F |  | Optical polarization evolution and transmission in multi-Ranvier-node axonal myelin-sheath waveguides [preprint] |  | 2023-(13) | Emily Frede Hadi Zadeh-Haghighi, Christoph Simon |
| F |  | Photons guided by axons may enable backpropagation-based learning in the brain |  | 2022-(11) | Parisa Zarkeshian, Taylor Kergan, Roohollah Ghobadi, Wilten Nicola, Christoph Simon |
| A |  | Electromagnetic modeling and simulation of the biophoton propagation in myelinated axon waveguide |  | 2022-(1) | Haomin Zeng, Yunhua Zhang, Yue Ma, Song Li |
| F |  | Engineering Photonic Transmission Inside Brain Nerve Fibers |  | 2021-(12) | Amir Maghoul, Ali Khaleghi, Ilangko Balasingham |
| F |  | Photons detected in the active nerve by photographic technique |  | 2021-(11) | Andrea Zangari, Davide Micheli, Roberta Galeazzi, Antonio Tozzi, Vittoria Balzano, Gabriella Bellavia, Maria Emiliana Caristo |
| F |  | A new viewpoint and model of neural signal generation and transmission: Signal transmission on unmyelinated neurons (terahertz/infrared) |  | 2020-(11) | Zuoxian Xiang, Chuanxiang Tang, Chao Chang, Guozhi Liu |
| F |  | Amplification of terahertz/infrared field at the nodes of Ranvier for myelinated nerve (terahertz/infrared) |  | 2020-(4) | Yan Sheng Liu, Kai Jie Wu, Chun Liang Liu, Gang Qiang Cui, Chao Chang, Guozhi Liu |
| A |  | A primary model of THz and far-infrared signal generation and conduction in neuron systems based on the hypothesis of the ordered phase of water molecules on the neuron surface I: signal characteristics (unmelyneated axons, terahertz/infrared) |  | 2020-(1) | Zuoxian Xiang, Chuanxiang Tang, Chao Chang, Guozhi Liu |
| A |  | Electromagnetic Waves Guided by a Myelinated Axon in the Optical and Infrared Ranges |  | 2019-(1) | O. M. Ostafiychuk, V. A. Es'kin, A. V. Kudrin, A. A. Popova |
| F |  | Electromagnetic Propagation Models in Nerve Fibers (myelinated axons) [conference] |  | 2019-(4) | Qingwei Zhai, Kelvin J. A. Ooi, C. K. Ong, Shengyong Xu |
| F |  | On the delay in propagation of action potentials (myelinated and unmelyneated axons) [preprint] |  | 2019-(18) | J. Xu, S. Xu, F. Wang, S. Xu |
| F |  | Cell vibron polariton in the myelin sheath of nerve (myelinated axons) [preprint] |  | 2019-(16) | Bo Song, Yousheng Shu |
| A |  | Myelin Sheath as a Dielectric Waveguide for Signal Propagation in the Mid-Infrared to Terahertz Spectral Range (myelinated axons, terahertz/infrared) |  | 2018-(1) | Guozhi Liu, Chao Chang, Zhi Qiao, Kaijie Wu, Zhi Zhu, Gangqiang Cui, Wenyu Peng, Yuzhao Tang, Jiang Li, Chunhai Fan |
| F |  | Node of Ranvier as an Array of Bio-Nanoantennas for Infrared Communication in Nerve Tissue(myelinated axons, terahertz/infrared) |  | 2018-(19) | Andrea Zangari, Davide Micheli, Roberta Galeazzi, Antonio Tozzi |
| F |  | Are there optical communication channels in the brain? (myelinated axons) |  | 2018-(15) | Parisa Zarkeshian, Sourabh Kumar, Jack Tuszýnski, Paul Barclay, Christoph Simon |
| F |  | Possible existence of optical communication channels in the brain (myelinated axons) |  | 2016-(24) | Sourabh Kumar, Kristine Boone, Jack Tuszýnski, Paul Barclay, Christoph Simon |
.
.